Why Modern Clinical Research Blends Site-Based Rigor with Digital Flexibility
Table of Contents
Clinical trials are undergoing a structural transformation. What began as an emergency response during the COVID-19 pandemic has now evolved into a lasting operational shift across the life sciences industry.
By 2026, more than 70% of clinical trials incorporate decentralized elements, combining traditional site visits with remote patient participation through digital tools. Fully decentralized clinical trials (DCTs), once seen as the future of research, remain relatively rare due to regulatory oversight requirements, data integrity considerations, and operational complexity.
Instead, the industry has converged around a more practical model: hybrid clinical trials.
Hybrid trials blend the scientific rigor of site-based research with the flexibility of decentralized technologies—allowing patients to participate from home while ensuring investigators maintain oversight for critical procedures.
For sponsors and CROs, hybrid models offer the best of both worlds: improved patient accessibility, streamlined operations, and regulatory confidence.
But hybrid trials also introduce new operational demands. Managing distributed data streams, remote monitoring, and patient engagement across multiple environments requires a unified digital backbone.
This is where platforms such as Mushroom Solutions’ CTOps and decentralized trial infrastructure play a crucial role—connecting clinical sites, patients, and data into a single operational ecosystem that supports hybrid trial execution at scale.
Why Fully Decentralized Trials Remain Rare
Despite the excitement surrounding decentralized research, fully remote clinical trials remain uncommon in practice. Several structural realities explain why.
Regulatory Oversight Still Requires Sites
Regulators continue to require investigator supervision for many critical trial endpoints. Complex diagnostics, imaging procedures, biopsies, and investigational drug administration often must occur within controlled clinical environments.
Site visits remain essential to ensure patient safety, protocol adherence, and validated data collection.
Data Integrity Challenges Persist
Purely remote trial models introduce potential variability in data capture. Wearable devices, home assessments, and patient-reported outcomes must be validated and reconciled with clinical data sources.
Without robust oversight and integration, decentralized data streams can introduce inconsistencies that complicate regulatory submissions.
Patient and Site Preferences Favor Hybrid Models
Patients often prefer the reassurance of periodic site visits, particularly for serious or complex therapeutic areas. Investigators similarly value in-person interaction for clinical judgment and patient monitoring.
Hybrid models offer the flexibility of remote participation while maintaining trusted site relationships.
Regulatory Guidance Encourages Balanced Approaches
CMC documents frequently undergo dozens of revisions during development. Without centralized version coRecent regulatory guidance has increasingly emphasized risk-based decentralization, where appropriate trial activities occur remotely while critical procedures remain site-based.
As a result, hybrid trial adoption has surged, balancing innovation with compliance.
Rise of Hybrid Clinical Trial Models
The shift toward hybrid trials accelerated rapidly following the pandemic, as sponsors sought ways to maintain study continuity while reducing patient burden.
By 2026, hybrid models have become widely recognized as the operational standard for modern clinical research.
Post-Pandemic Acceleration
The rapid adoption of telemedicine, remote monitoring, and digital data collection during the pandemic demonstrated that many trial activities could occur outside traditional sites.
Sponsors quickly realized that decentralization could improve recruitment and patient engagement without compromising study integrity.
Efficiency Gains
- Patient retention increases of up to 25%
- Operational cost reductions of up to 30%
- Faster recruitment timelines
- Improved patient diversity and geographic reach
These improvements stem largely from reduced travel burden and increased accessibility for participants.
Global Trial Expansion
Hybrid infrastructure has also enabled sponsors to run more globally distributed trials.
Today, more than half of clinical trials take place outside the United States, reflecting the industry’s move toward broader patient populations and international collaboration.
Hybrid models make this global expansion feasible by enabling remote patient monitoring and digital trial coordination across regions.
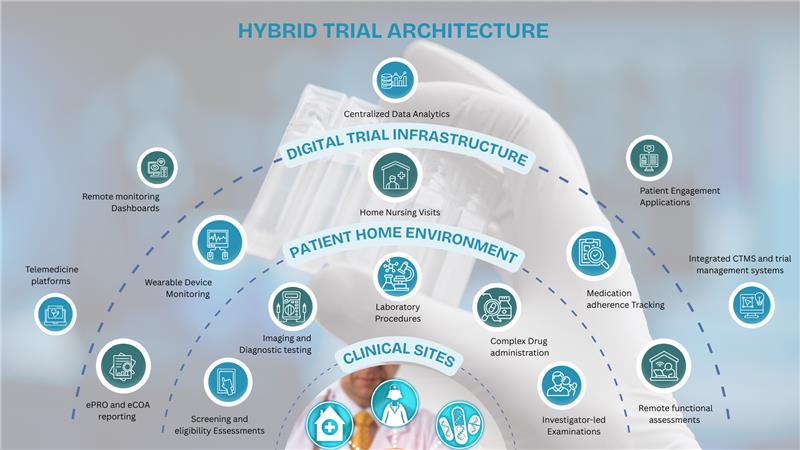
Operational Frameworks for Hybrid Clinical Trials
While hybrid trials improve patient accessibility, they also require careful operational design. Sponsors must determine which trial activities should occur at sites and which can safely occur remotely.
Successful hybrid trials rely on structured frameworks that align clinical procedures, technology platforms, and operational workflows.
Design Principles for Hybrid Trials
Activity Mapping
One of the most important steps in hybrid trial design is mapping activities to their optimal environments.
Certain procedures remain best suited for clinical sites:
- Imaging and diagnostics
- Biopsies and lab assessments
- Investigator-led examinations
- Drug administration requiring supervision
Other activities can easily occur remotely:
- Telemedicine consultations
- Wearable device monitoring
- Home nursing visits
Mapping these activities appropriately ensures both patient convenience and data reliability.
Technology Infrastructure
Hybrid trials depend on integrated technology ecosystems that support multiple modes of participation.
This includes:
- Virtual site platforms
- Digital patient engagement tools
- Remote monitoring systems
- Centralized analytics dashboards
- Regulatory-compliant data architectures
Without integration, hybrid trials risk fragmentation across multiple vendors and data silos.
Key Components of Hybrid Trial Operations
The following framework illustrates how hybrid trials distribute responsibilities across sites, patients, and digital platforms.
| Framework Element | Site Role | Home/Digital Role | Mushroom Enablement |
| Recruitment & Screening | Initial patient visits and eligibility assessments | eConsent platforms and digital pre-screening tools | CTOps recruitment automation and patient engagement workflows |
| Data Capture | Investigator-led eSource entry and CRF documentation | Remote ePRO/eCOA reporting and functional performance uploads | eCOA/ePRO validation and ePerfo remote performance data capture |
| Monitoring & Safety | Site oversight and risk-based monitoring reviews | Wearable devices and remote safety reporting | CTMS dashboards and real-time RBQM analytics |
| Logistics & Drug Supply | Site inventory management and drug dispensing | Direct-to-patient delivery and adherence monitoring | Automated supply chain workflows and operational SLAs |
Governance and Data Interoperability
Hybrid trials require strong governance models to ensure data consistency across distributed sources.
Modern frameworks increasingly adopt interoperability standards such as:
- USDM (Unified Study Definitions Model)
- DDF (Digital Data Flow)
These frameworks enable standardized data exchange across digital platforms, sites, and regulatory systems.
Pilot programs increasingly measure hybrid trial performance using metrics such as data freshness, monitoring responsiveness, and patient engagement levels, with some platforms achieving near real-time data visibility across trial environments.
Mushroom Solutions as a Hybrid Trial Enabler
Executing hybrid trials successfully requires more than individual tools. Sponsors need a unified operational platform that connects patient participation, clinical site workflows, and regulatory data management.
Mushroom Solutions provides this infrastructure through its CTOps and decentralized trial platform.
The platform supports many of the key capabilities required for hybrid trials, including:
- Hybrid site and remote patient workflows
- ePRO and eCOA digital patient reporting
- eSource-enabled clinical data capture
- Real-time monitoring and analytics
- Regulatory-aligned data governance
Across the broader decentralized trial landscape, Mushroom supports many of the core operational trends shaping modern clinical research, including hybrid trial execution, digital patient engagement, remote monitoring, and regulatory-compliant data management.
Differentiation in the Decentralized Trial Ecosystem
While several vendors focus on fully decentralized trial technologies, Mushroom emphasizes operational integration and automation.
This approach provides several advantages:
- Reduced platform fragmentation
- Cost-efficient implementation for mid-size sponsors
- Alignment with modern interoperability standards
- Automation across trial operations
Example: Remote Performance Monitoring
One example of hybrid enablement is ePerfo, which allows patients to perform functional assessments from home—such as walking tests or grip strength evaluations—while automatically uploading performance data into the trial platform.
By capturing validated performance metrics remotely, sponsors can reduce site visits while maintaining high-quality clinical data.
This approach helps improve patient adherence and reduce dropout risk while ensuring investigators maintain oversight of study outcomes.
Strategic Value of Hybrid Trial Infrastructure
Hybrid clinical trials are no longer experimental—they are becoming the operational baseline for modern clinical research.
Organizations that successfully implement hybrid models can realize several strategic advantages:
- Faster trial startup timelines
- Improved patient recruitment and retention
- Reduced operational costs
- Expanded global participation
- stronger regulatory readiness
However, achieving these outcomes requires integrated infrastructure capable of managing distributed clinical environments.
Platforms like Mushroom Solutions’ CTOps enable sponsors to design hybrid trials that combine digital flexibility with regulatory confidence.
Conclusion
The clinical research industry is moving beyond the binary choice between traditional site-based trials and fully decentralized models.
The future lies in intelligent hybrid designs that combine the strengths of both approaches.
Hybrid trials allow investigators to maintain clinical rigor while bringing research closer to patients—improving accessibility, operational efficiency, and study outcomes.
As digital technologies, data standards, and regulatory frameworks continue to evolve, hybrid models will increasingly become the default architecture for clinical trials across all phases of research.
Organizations that invest in integrated hybrid trial infrastructure today will be best positioned to run faster, more efficient, and more inclusive studies tomorrow.
With unified operational platforms like Mushroom Solutions’ CTOps, sponsors can transform hybrid trials from a logistical challenge into a strategic advantage.


